 Isoelectric focusing is widely applied in the determination of isoelectric points of protein, as well as protein separation. Storing it dry after eliminating background. Staining: remove the plastic pieces, place into the 65 ℃ dye solution until bands appear.ĩ. Fix: remove the plastic sheet, placing into fixative for 15min.Ĩ. Determination of isoelectric point of proteinħ. And then stop it once the electric current reaches 4mA.Ħ. And then continuing electrophoresis for 30 minutes. 
After 10 minutes, removing paper and stopping electrophoresis. Closing the lid, and performing electrophoresis with 25W. Placing a lens mirror paper in any position on the surface of glue. And then putting an electrode strip impregnated with electrode buffer on both sides of plastic surface. Placing the plastic gasket with clean bottom on the electrophoresis tank. Uncovering the up and down glass sheets after the gel coagulation. And then clamping the glass sheets with good position. Performing two clean special IEF glass with suicide and anti-suicide reaction. Sample Buffer: 1% Ampholine, 2% Triton X-100,9M ureaĮxtracting pending samples with IEF sample buffer. Staining solution: 0.35g Coomassie brilliant blue R-150 was dissolved in 300ml destaining solution and heated to 60-70 ℃, copper sulfate was added 0.3gĭestaining solution: 25% ethanol, 8% acetic acid is dissolved in water Polyacrylamide, B polyacrylamide, amphoteric electrolytes, urea, NP-40, teiton-100Įlectrode solution: 1M phosphoric acid (anolyte), 1M sodium hydroxide (catholyte)įixative: 100g TCA, 10g sulfosalicylic acid dissolved in 500ml, a total volume of 1000ml And because of the change of pH, molecules will get a positive or negative charge, as long as they are away from their isoelectric points. Simultaneously, molecules focusing on isoelectric points will also spread. And then the pH can be regarded as the pI of this molecule. Protein molecules or other amphiphilic molecules in such pH gradient will reach a zone with zero static charge due to the movement of their surface charge in this electric field. When the electric field is applied, a stable pH gradient between the poles will be established as a result of the movement of amphiphilic carriers. The salt addition method has been found appropriate and convenient to determine the PZC of natural organic substrates.Ĭation exchange capacity Ion adsorption method Isoelectric point Organic materials Point of zero charge Point of zero net charge Salt addition method Zeta potential.Isoelectric focusing(IEF), performing a very important role in protein separation technology, is utilized to separate amphoteric macromolecules in line with the different isoelectric points.įilling the electric field with amphiphilic carriers and anti-convection media. As for the goethite, the PZC (10.9 ± 0.05) was slightly higher than the PZNC (9.0-9.4). The PZC of kaolinite (3.01 ± 0.03) was similar to the PZNC (2.9-3.4) and fell within the range of values reported in the literature (2.7-4.1). In this work, isoelectric points (p I s) of 23 Food and Drug Administration (FDA) and European Medicines Agency (EMA) approved mAbs were determined by imaged capillary isoelectric focusing (icIEF), and ranged from 6.1 to 9.4. Both the ion adsorption and the zeta potential methods failed to give points of zero charge for these substrates. Results obtained from the salt addition method in 0.05 M NaNO 3 were the following: 4.72 ± 0.06 (maple sawdust), 9.50 ± 0.07 (wood ash), 3.42 ± 0.03 (peat moss), 7.68 ± 0.01 (green compost), and 6.06 ± 0.11 (brown algae). Natural kaolinite and synthetic goethite were also tested with both the salt addition and the ion adsorption methods in order to validate experimental protocols. Three methods were used: (1) the salt addition method, measuring the PZC (2) the zeta potential method, measuring the isoelectric point (IEP) (3) the ion adsorption method, measuring the point of zero net charge (PZNC). The PZC provides important information about metal sorption mechanisms. 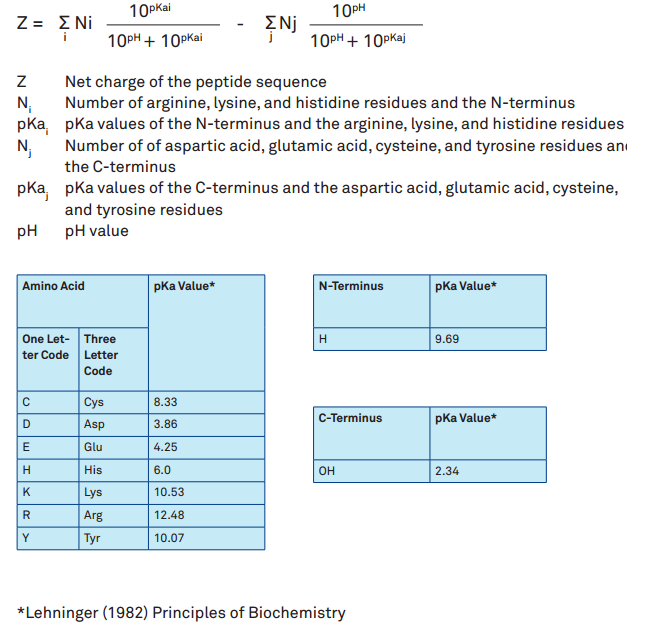
This study evaluates different methods to determine points of zero charge (PZCs) on five organic materials, namely maple sawdust, wood ash, peat moss, compost, and brown algae, used for the passive treatment of contaminated neutral drainage effluents. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
